Cryogenic transmission electron microscopy (cryo-TEM) enables researchers to work out the structure of the proteins, viruses and other molecular complexes that underlie all of life’s processes – and its diseases. In particular, the technique is critical to global health research and drug discovery.
The microscopes needed to get the highest resolutions are extremely expensive and complex, largely due to their high-energy (300keV) electron sources. They sit firmly at the cutting edge of instrument design and operation, often costing around $10M. Even a ‘standard’ cryo-TEM that is used for the screening part of the structure-determination process is $4–5M. This often makes cryo-TEM prohibitively expensive, particularly in resource constrained institutions and nations. A more equitable solution would help to uplift the application of this powerful technology.
A team at Microscopy Australia’s Monash University facility – Ramaciotti Centre for Cryo-Electron Microscopy – has demonstrated that with the addition of a new detector and cryogenic sample holder, a significantly cheaper “standard” TEM can achieve a seven-fold improvement in resolution, enabling it to determine the structures of proteins.
In work supported by the Chan Zuckerberg Initiative and based on recent findings from Nobel Laureates Dr Richard Henderson and Dr Chris Russo at Oxford, Microscopy Australia Platform Scientist, Dr Hari Venugopal and Facility Director, Prof. Georg Ramm, at the Ramaciotti Centre for Cryo-EM, explored whether a standard TEM (120-keV) with the basic electron source can be upgraded for high-resolution cryo-EM. They showed that even this ‘ordinary’ TEM, found in many facilities around the world, when fitted with a new ultra-sensitive direct electron detector camera can achieve structures with a remarkably improved resolution over the same microscope in its traditional configuration. It had previously been assumed that higher energy electron beams were better for determining protein structures but this is not actually the case.
The ability to generate the high-resolution structure of a protein depends on the protein’s size, stability and symmetry, with the highest resolution structures being achieved from large, symmetrical proteins with a stable structure.
In a partnership with Gatan, the Monash team added a new, ultrasensitive Gatan camera to one of these basic microscopes and set about determining the structures of three different proteins in a variety of instrument configurations. They were able to obtain a 2.67 Å resolution structure for the large, stable, symmetrical protein apoferritin, compared to 1.4 Å on a top-end microscope. For the smaller symmetrical protein, haemoglobin, they achieved 4.33 Å resolution compared to 2.3 Å from on a top-end microscope. As well as these two commonly used standards, they also applied the technique to an asymmetric, flexible, medium-sized protein, GPCR, part of a protein family relevant for many drug targets and which has been a difficult structure to solve. For this, they reached 4.4 Å resolution compared to 2.4 Å on a top-end microscope.
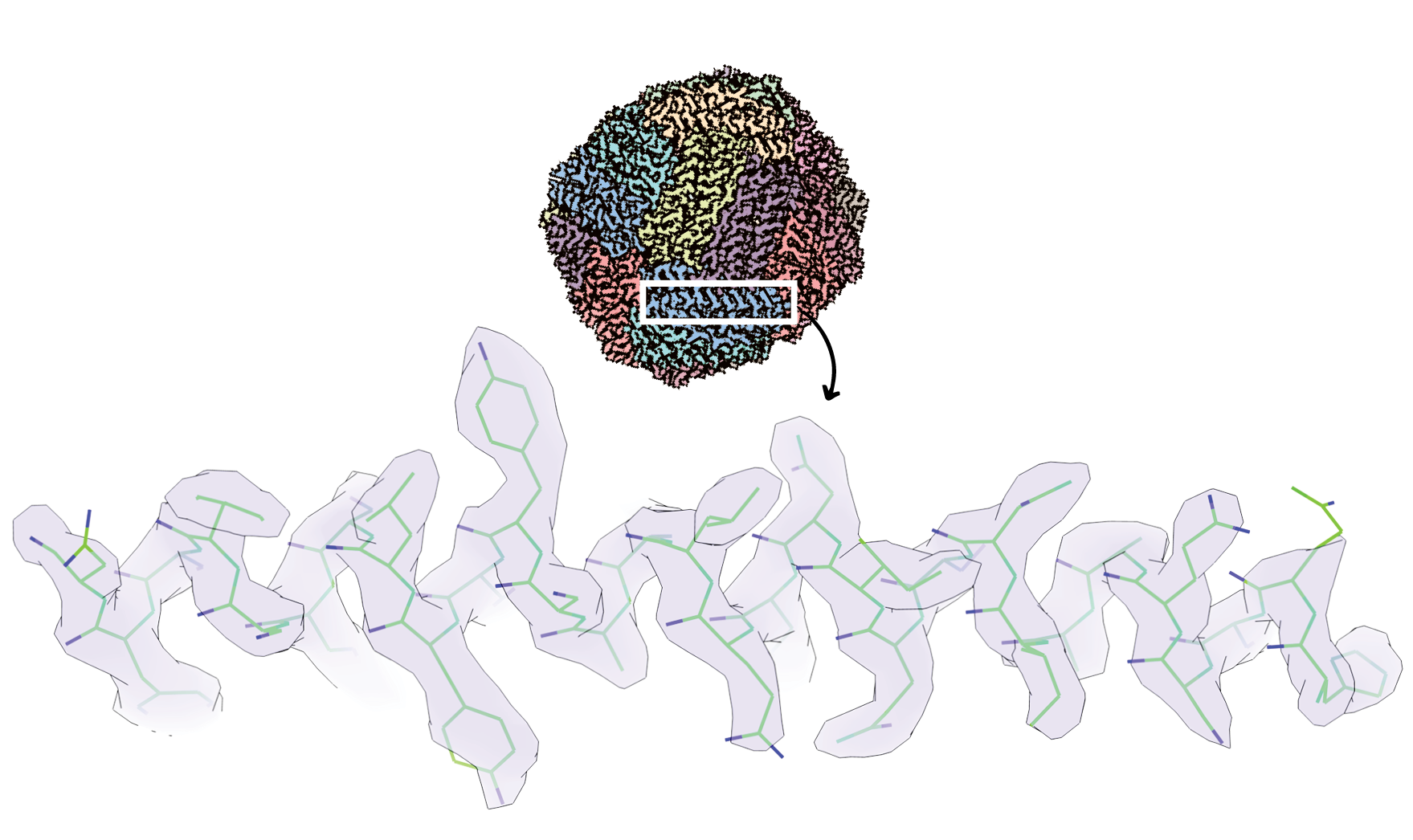
Revealing the resolution achieved: above is the whole protein apoferritin – the iron-storage component of ferritin, a crucial measure of anaemia – imaged using the new technique. Below, reveals the resolution when you zoom into this. Each bend, or end, of the green line indicates the location of a molecule or atom in the protein sub-unit, blue indicates a specific type of molecule.
Another feature of the work is that the team achieved those structures from collecting a relatively low number of individual molecules, also making the process relatively quick and inexpensive. These resolution gains could potentially be improved further with more individual molecules feeding into the final structure and further optimisation of the microscope configuration.
The team showed that the high resolution needed for preliminary protein structure determination can now be achieved at a fraction of the cost, contributing significantly to the democratisation of cryo-TEM. This kind of set up will never be able to achieve resolutions better than 2 Å due to the physical limits of the system and so will not replace the current high-end microscopes. Nevertheless, it has shown what is possible and is a huge step forward in making cryo-TEM a more affordable technique and opening it up to a much larger global research community as well as reducing costs for everyone.
H. Venugopal et al. Science Advances 2025
DOI: 10.1126/sciadv.adr0438
These structures were captured on equivalent microscopes. The lower is a phage – a virus that attacks bacteria – captured in 2012, and the one above is apoferritin – the iron-storage component of ferritin – captured in 2025 with the upgraded detector. The improvement in resolution is striking: 2 nm to 0.265 nm.
May 5, 2026