The nano-sized cages, called encapsulins, act like miniature containers in the cell, housing enzymes that conduct essential functions. Encapsulin cages are made from simple proteins that spontaneously self-assemble into porous icosahedral structures, which work by clustering enzymes together into dedicated confined spaces. This proximity gives them the right environment to work properly.
These cages are highly effective at providing a space for many of the chemical reactions that take place inside cells, allowing some substances to pass into the interior while not letting toxic intermediates get out and damage the cell. Taking the lead from nature, chemists and synthetic biologists alike want to engineer bespoke compartments to house different chemical reactions. This would create nanoreactors capable of producing a wide array of important molecules that could otherwise harm the cells if they floated freely.
An article published in the journal Science Advances, reports the teams’ work and reveals how the porosity of encapsulins controls the way that valuable molecular products can enter and leave these cages. The research team led by Dr Yu Heng Lau, from the University of Sydney, designed and solved the structures of a series of synthetic encapsulins, designed to have different porosities.
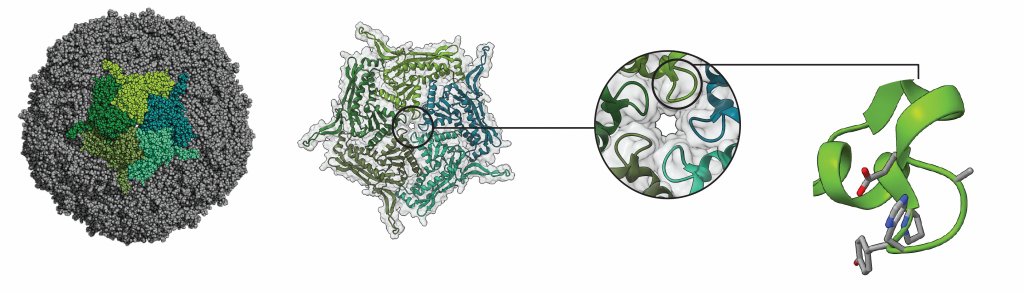
24 different variants of an encapsulin were engineered by the researchers and, by using cryo-TEM and the associated expertise in the Microscopy Australia facilities at UNSW Sydney and the University of Sydney, they showed how the structures of some of these variants differed from each other. They were then able to work out how these differences affect the way the cages function. They found that a combination of pore size and electric charge was responsible for controlling how well different molecules move in and out of the cages.
These potential nanoreactors are a tool with great promise and this study has started writing the rule book to guide and enhance design of future nanoreactors.
L. Adamson et al. 2022 Science Advances DOI: 10.1126/sciadv.abl7346
Structures of engineered nanoreactor pores revealed by cryo-EM. Red shows negatively charged regions and blue, positively charged regions.
February 7, 2022