The research, lead by Dr Abdalla Ali from the University of Queensland School of Dentistry, builds on existing scaffolds used for bone regeneration made from a polymer called polycaprolactone (PCL) by adding bio-active magnesium hydroxide. The addition of the magnesium hydroxide was shown to enhance the metabolic activity, attachment, and proliferation of bone forming cells, and helps the scaffold to degrade more quickly which is desirable for bone formation.

Scanning electron microscope image of bone scaffold showing interconnected pore space.
Scanning electron microscopy at CARF allowed researchers to confirm that pores in the scaffold were interconnected. Interconnected pore space has been shown to positively influence cell growth rate and depth of migration into bone scaffolds by providing space for the vasculature required to nourish new bone formation and remove waste metabolites. In addition, EDS, a type of elemental mapping, was used to confirm the incorporation of magnesium hydroxide in the scaffold.
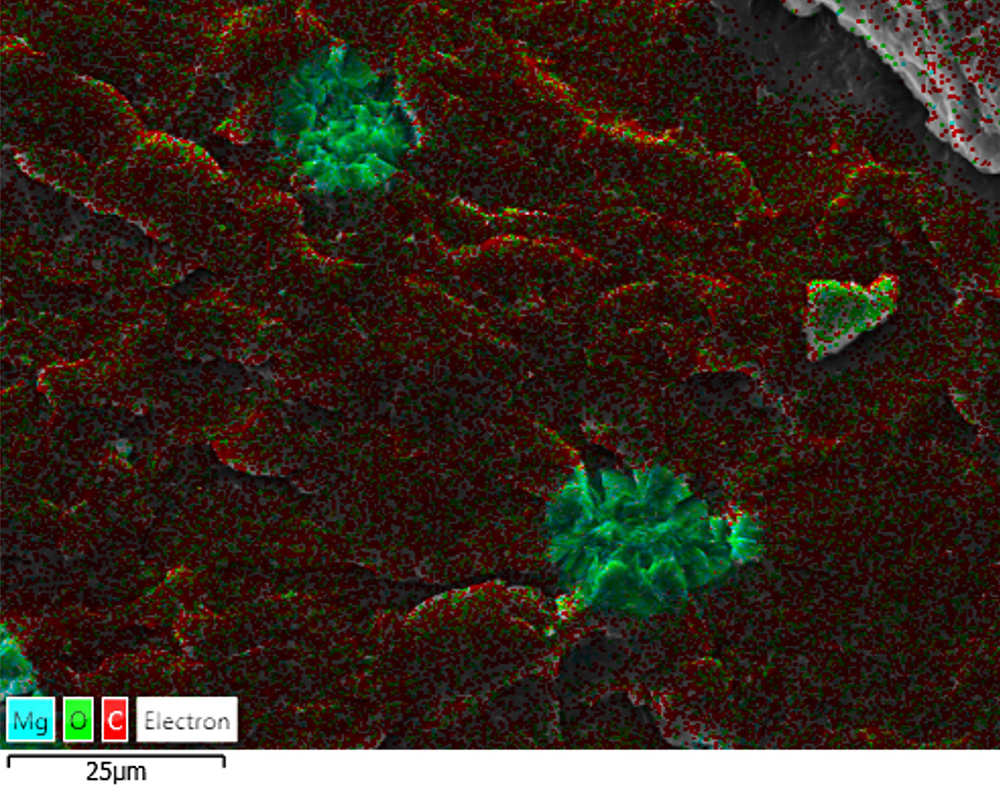
Scanning electron microscope image with coloured EDS (elemental analysis) overlain
Dr Ali said human bone grafts were considered the traditional ‘gold standard’ for restoring damaged or injured bone that was not capable of healing itself. “But bone grafts have the drawback of limited availability, and are subject to complications. 3D-printed bone scaffolds are a new alternative made from porous degradable biomaterials that provide mechanical support during the bone’s natural repair process,” he said.
“PCL has a slow degradation rate that is not ideal for many bone healing applications and lacks biofunctionality. Many attempts have been made to incorporate new materials into bone tissue engineering, but they are not fully absorbable in the body, which is necessary as the bone naturally regenerates.”
Dr Ali said adding magnesium hydroxide in their study of 3D-printed scaffolds enhanced bone formation, resulted in a higher bone mass and a faster degradation rate.
Bone implant rates are rising globally due to the increasing ageing population, leading to more age-related degenerative bone problems, bone diseases, accidents and injuries, and revision surgery.
Scanning electron microscope image of the bone scaffold
January 13, 2021